
Many consumers do not know that Polystyrene is the number six plastic and is represented
by the above symbol:
Polystyrene is found in your home, office, local grocery and favourite restaurant and it comes
in many shapes and forms. Polystyrene provides strength, lightness, durability, heat resistance, transparency, gloss, desirable appearance and practical toughness for a wide range of uses, including packaging, medical products, appliances and electronic products.
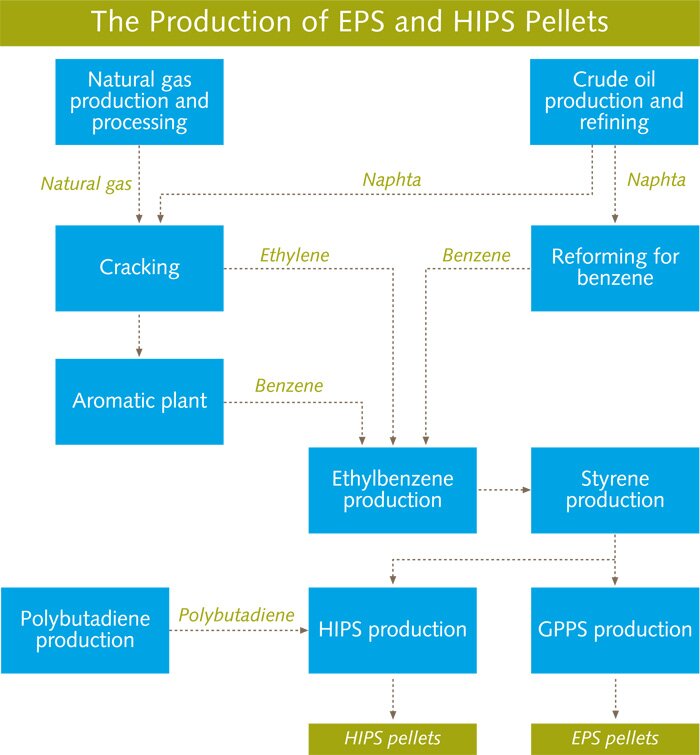
Herewith the two most widely used forms of Polystyrene
In 1839 Polystyrene was discovered, but was only made practical in 1938. In 1954 Styrofoam, the trademarked form of Polystyrene foam insulation, was invented.
Expanded polystyrene is made from expandable polystyrene, which is a rigid cellular plastic containing an expansion agent. EPS is obtained from oil and the raw material for EPS is produced in the form of small polystyrene beads containing a blowing agent, or pentane, which when exposed to steam, expand to form a light weight "pre-foam" of required density. This pre-foam is then processed by further steam treatment until the beads fuse together, either in a mould to give that material a required shape and size or as large blocks for cutting into sheets and shapes.
Is EPS a widely used plastic?
Yes. EPS is among the biggest commodity polymers produced in the world. The total world demand in 2001 was 3.06 million tons and is expected to grow at 6 percent per year. EPS is
a solid foam with a unique combination of characteristics, like lightness, insulation properties, durability and an excellent process ability. EPS is used in many applications like thermal insulation board in buildings, packaging, cushioning of valuable goods and food packaging.
Go to our Downloads page for more scientific articles on the manufacturing of Polystyrene
Uses of EPS
Any product of any shape or size may be packaged in expanded polystyrene. The following
only touches on the possibilities in both the industrial and food packaging sectors using EPS.
- Food packaging, only to name a few:
-
- Hot beverage cups
- Vegetable trays
- Meat trays
- Hamburger clam shells
- Protective and display packaging:
-
- Around new electronic equipment for example televisions and hi-fi’s
- Protecting new appliances such as refrigerators, washing machines etc.
- Building and Contruction (EPSASA)
- Other applications
Apart from the typical application in construction and packaging, EPS protective qualities can also be used in crash helmets - protecting the heads and potentially the lives of cyclists, or into surface and other decoration ranging from simple printing of a brand name to an elaborate pictorial representation achieved by mould engraving, or for fun and sports e.g. windsurfing board.
Polystyrene is the fourth biggest polymer produced in the world after polyethylene, polyvinyl chloride and polypropylene.
General purpose polystyrene (GPPS) is a glasslike polymer with a high processability. When modified with rubber it results in a high impact polystyrene (HIPS) with a unique combination
of characteristics, like toughness, gloss, durability and an excellent processability.
Polystyrene is one of the most versatile plastics.
Whether packaging for food products, in office and information technology or refrigerators,
all sectors place high demands on the properties of the materials used. In its diverse variants HIPS offers extraordinary property combinations, thus making a vital contribution to everyday life. High impact polystyrene is also used in many applications because of its excellent balance of properties and low cost.
HIPS also have good impact resistance, good dimensional stability, excellent aesthetic qualities, is easy to paint and glue, can be manufactured at a low cost and is approved by the U.S. Food and Drug Administration.
Typical applications include food service disposables, food packaging, appliance housings,
toys, electronic applications and industrial packaging.
- Food service disposables and -packaging, only to name a few:
-
- Yogurt tubs
- Salad bowls
- Disposable knives, forks and spoons
- Display and other packaging:
-
- Refrigerator linings
- CD cases
- Medical trays
The legislation for polystyrene (PS), expandable polystyrene (EPS), and Acrylonitrile-Butadiene-Styrene copolymer (ABS) and Styrene-Acrylonitrile copolymers (SAN) as well as other plastics in contact with food within the EU is based on a system of self-certification. Polymers sold for use into food-contact applications are not assessed by any legal authority, nor is there official product registration of food approved polymers. Instead, each producer has the individual responsibility to ensure that each material, which he markets for use in contact with food, does comply with the applicable regulations, and to certify this to his customers.s.
The key elements of the European legislation regarding food-contact plastics are the following:
1. Compositional requirements
- The components used in the manufacturing of the polymer need to be registered
on a 'positive list'.
- The main components needed for the production of a polymer, namely the monomers and additives, are regulated by means of EU Directives. The provisions of these Directives apply to all EU Member countries, and are enforced in the national laws.
- In addition to the above, the following countries still have country-specific restrictions regarding the composition of the material: Germany, the Netherlands, Belgium,
France, Italy and Spain that address aspect not yet resolved by the EU directives
(eg. colorants, catalysts, etc.).
Note: also the USA and Japan have their regulation on food contact materials.
2. Migration Limits
In order to safeguard the inertness of the plastic towards the food, the migration of volatile substances (such as additives and residual monomers) from the food contact article into the food needs to meet prescribed migration limits.
For styrene polymers the migration on the finished articles should not exceed the following limits:
- overall migration less than 10 mg per dm˛ of the article surface,
- for high impact polystyrene butadiene migration must be "non-detectable" (detection limit of method 0.02 mg/kg food) or alternatively, residual butadiene content in the finished article must be less than 1 mg/kg.
- for ABS (beside the mentioned restriction on residual butadiene) and SAN, acrylonitrile migration into food must also be "non-detectable" (detection limit of method 0.02 mg/kg food). Differently from butadiene, in this case it is not allowed to decide compliance based on the acrylonitrile residual in the polymer.
- for certain ingredients additionally specific migration limits (SML) may be imposed since the publication of Commission Directive 2001/62/EC, the actual migration should not exceed these limits.
No migration limit is in place for styrene monomer.
The conditions under which the migration has to be tested is prescribed in the Commission Directive 97/48/EC of 29 July 1997. As it is not always possible to use foodstuffs directly for testing, food simulants are introduced. These are:
| Aqueous food (pH > 4.5) |
Distilled water or equivalent |
| Acidic foods (pH < 4.5) |
3 percent acetic acid in water |
| Alcoholic foods |
10 percent ethanol in water, or actual alcoholic strength if concentration exceeds 10 percent |
| Fatty foods |
Rectified olive oil, or other fatty food simulants, meeting certain specifications, e.g. HB 307 |
The contact time and temperature depend on the application that has to be tested. The standardised contact time may range from 30 minutes till 10 days, and the temperature may range from 5 °C up to 175 °C, also depending on application and polymer type. For polystyrene polymers usually 2 hours/70° C and 10 days/40 °C is used. For repeated use articles (such as kitchenware) the migration testing is to be made three times on the same article, using only the last extract for the migration measurements. This is important particularly for acrylonitrile, since the specific migration limit is low. The manufacturer of the finished article must check that the polymer specification will fully meet the technical requirements of the final article. Also the manufacturer of the finished food contact article must provide the final users with adequate instructions for use of these finished food contact articles, this includes instructions for consumers, in case these articles are sold for in-house application.
3. Off Taste
The taste of the food should not be unacceptably altered by the contact with the plastic.
4. Good Manufacturing Practices
The polymer must to be produced according to good manufacturing practices.
| EU |
Commission Directive 2002/72/EC containing the consolidated text of all previous versions to start with 90/128/EEC (applies to all EU Member States) and following amendments. |
| Germany |
BfR Recommendation VI "Bedarfsgegenständeverordnung" of 10-04-1992, and subsequent modifications. |
| Belgium |
Royal Decree 92-1952 of 11-05-1992, and subsequent modifications up to date. |
| France |
"Modified Brochure 1227 (Journal Officiel de la République Française du 15 July 2002)" and subsequent modifications. |
| Italy |
"Decreto Ministeriale" of 21-03-1973 and subsequent modifications. |
| Netherlands |
"Verpakkingen en Gebruiksartikelen Besluit" , Staatcourant 25 January 1980, nr. 18 including subsequent modifications. |
| Spain |
"Real Decreto" RD 118/2003 and subsequent modification in Orden SCO/983/2003. For additives Resolution 4-11-82.. |
| USA |
Food, Drug and Cosmetic Act as amended under Food Additive Regulation 21 CFR §:
177.1020 for ABS copolymers/repeated use,
177.1640 for GPPS and HIPS
177.1810 for Styrene block polymers and
181.32 for Acrylonitrile copolymers
The use cited above is subject to good manufacturing practices and any limitations which are part of the regulations (consult the appropriate regulations for complete details). |
| Japan |
Food sanitation law No 233 of 1974.
Self-restrictive requirements of food contacting articles made of polyolefins and certain polymers, 3rd Edition, September 1988, Japan Hygienic Olefin and Styrene Plastics. |
Product safety aspects of styrenic materials and their ingredients:
The styrenics are polymers that can be safely used in many applications. Although some of their ingredients may be dangerous, when used in the polymer they mostly do not present a danger to human health (for instance by inhalation, ingestion or contact with skin) since such ingredients are embedded in the polymer matrix.
- Styrene monomer
- Acrylonitrile monomer
- Brominated additives
- Blowing agents
Currently, based on the EU classifications, styrene monomer - the major building block of polystyrene - is considered to have low oral toxicity, and is not classified in terms of carcinogenicity or mutagenicity.
Styrene monomer is one of many large volume chemicals subject to review based on the European regulation EEC/793/93, which requires chemical risk assessments. This process evaluates both the hazard (e.g. potential for causing health effects) of a substance and the real-life potential for exposure to that substance (e.g. in the environment, workplace, etc.)
to determine whether or not there might be a risk from exposure to a substance under certain conditions. The United Kingdom's (UK) Health and Safety Executive (HSE) has drafted this risk assessment, which is under review by EU experts , and will be the basis for reviewing the classification of the product, its labelling requirements, and also regulations concerned with worker exposure.
PlasticsEurope’s Polystyrene and Expandable Polystyrene Committee's work closely together within the styrenics chain of PlasticsEurope-CEFIC and with the e International Styrene Industry Forum (ISIF), whose mission it is to collect, develop, analyse and communicate pertinent information on styrene. Since 1987 a comprehensive research program has undertaken to enhance understanding of styrene's potential to affect human health and the environment.
Acrylonitrile monomer – one of the monomers used for the production of ABS and SAN
is classified as a category 2 carcinogen by the European Union.
Although, it is a confirmed carcinogen in animal tests, there is no proof that it triggers cancers in humans.
IARC (the International Agency for the research on Cancer) deem that “there is inadequate evidence in humans for the carcinogenicity of acrylonitrile”: the available studies do not permit a conclusion regarding a link between exposure to acrylonitrile and cancer in humans. Based on the evidences, while acrylonitrile is an animal carcinogen, the risk to humans is regarded to be low. This conclusion is also supported by the epidemiological studies available.
The European Acrylonitrile risk assessment was completed in 2001. This process evaluates both the hazard (e.g. potential for causing health effects) of a substance and the real-life potential for exposure to that substance (e.g. in the environment, workplace, etc.) to determine whether or not there might be a risk from exposure to a substance under certain conditions.
The EU Authorities recognised that even though a risk cannot be excluded, this is regarded to be very low for consumers and that the legislation already in place in Europe for acrylonitrile protects the consumer’s health. The risk assessment also clarified that the current workplace exposure limits in place for Acrylonitrile, represent a satisfactory precautionary measure to protect acrylonitrile-exposed workers.
The brominated substances used for fire ignition in expandable polystyrene and in some ignition resistant high impact polystyrenes have very different properties and most are of low toxicity. The most important brominated substances are subject to review based on the European regulation EEC/793/93, which requires chemical risk assessments. This process evaluates both the hazard (e.g. potential for causing health effects) of a substance and the real-life potential for exposure to that substance (e.g. in the environment, workplace, etc.) to determine whether or not there might be a risk from exposure to a substance under certain conditions. The Industry is actively promoting the production of scientifically sound data to support the risk assessment process. For further information consult the website of the European Brominated Flame Retardants Industry Panel, EBFRIP.
The very few references available indicate that pentane is not a toxic hazard from occupational exposure. Pentane has also low neuromuscular toxicity. There is also no evidence for carcinogenic, mutagenic or teratogenic activity from pentanes. Furthermore pentane is approved without restrictions by the European Union as additive to be used in plastics that come in contact with foods. Nevertheless, no pentane is expected to residue in an EPS-based final article.
CO2 is natural occurring gas, it is non-toxic, non-flammable, does not contribute to tropospheric smog, and has no stratospheric ozone depletion potential. In addition, the carbondioxide used for this technology is recovered from existing commercial and natural sources. As a result, the use of this blowing agent technology does not increase the levels of CO2 in the atmosphere.
Environment preservation
Climate change
Energy reduction
As excellent thermal insulator EPS dramatically reduces building energy requirements:
a well insulated 'detached one family house' demands only 30% of the heating energy t per year that a poorly insulated habitation requires (www.eumeps.org). EPS conserves fossil fuels
and cuts the carbon dioxide emissions associated with power generation (which add to the
greenhouse effect ) by about 300 000 kg over 50 years compared to a poorly insulated house..
Ozone depletionon
The EPS production poses no threat to the ozone layer: , the blowing agent used in EPS contains no ozone-depleting substances and the amount released during manufacture is
only about 0.2 percent of the total man-made emissions of volatile organic compounds (VOC's). It actually is a naturally occurring chemical like methane. It is also continually being formed
by natural processes; for example in the digestive tracts of animals and by the anaerobic decomposition of plant matter. It has a low stability and under the influence of humidity
and atmospheric radiation (specially UV light) it is quickly converted into carbon dioxide and water. Because of its low stability, pentane cannot reach the higher (stratospheric) atmospheric levels and therefore does not contribute to ozone depletion (unlike chlorinated blowing agents such as CFC's and HCFC's). Additionally pentane does not contribute directly to the "greenhouse" effect.
The table below illustrates the atmospheric effects of some chemical compounds and the
low significance of pentane:
| CFC11 |
1.0 |
0 |
65 years |
| HCFC 141b |
0.1 |
0 |
7.8 years |
| n-pentane |
0 |
40 |
8 days |
| n-butane |
0 |
40 |
9 days |
| methane |
0 |
1 |
7.1 years |
| CO2 |
0 |
0 |
50-200 years |
| O3 (troposphere) |
- |
- |
2 months |
1 ODP denotes Ozone Depletion Potential (by definition, ODP of CFC11 is 1.0)
2 POCP denotes Photochemical Ozone Creation Potential (by definition ethylene is 100)
With ever evolving technology, some manufacturers of extruded foam polystyrene - XPS for short - for packaging and insulation foam use carbon dioxide as a blowing agent. CO2 does not contribute to tropospheric smog and has no stratospheric ozone depletion potential. In addition, the CO2 used for this technology is recovered from existing commercial and natural sources. As a result, the use of this blowing agent technology does not increase the levels of CO2 in the atmosphere.
A product's environmental impact cannot accurately be determined simply by focusing on its disposal at the end of its life. A cradle-to-grave approach should be adopted, assessing its impact from manufacture through to final recovery or disposal. This approach, a developing science known as eco-profile or life-cycle analysis, allows objective decisions to be reached - ensuring maximum environmental protection when products are selected.
Eco-balance and life cycle analysis studies have illustrated that EPS offer environmental benefits over alternative materials in insulating products as well as in packaging.
Additionally, because of its excellent insulating properties and light weight, EPS actually saves natural resources during its lifetime:
- EPS insulation board means that less heating fuel - or energy for air conditioning - is required: insulating to the most stringent current regulations can cut heat loss by more
- than 70%, compared with a similar, non-insulated building. For every kg of oil used to manufacture an EPS insulation board, about 150 kg is saved over its functional (ca. 50 years) lifetime through reduced heating demands.
- EPS' excellent insulating properties mean that hot food stays hot and cold food cold for a longer period of time, reducing the need to pump precious energy resources into respectively refrigeration or heating. Moreover you can meanwhile hold the package in comfort.
- EPS and XPS insulation boards insulate houses, protective packaging protects valuable shipments etc. but above all because of its light weight, (E)PS reduces fuel consumption during transportation - since less weight to move means less fuel to use.e.
Thermoplastic polymers including polystyrene, both polystyrene and expandable polystyrene can either be mechanically recycled into several applications or chemically converted into other feedstocks. Incineration of plastics as part of the municipal solid waste stream generates substantial amounts of energy, saving the use of alternative fuels. The heat of combustion of polystyrene polymers is approx. 41 MJ/kg, comparable to the value for oil. In a proper designed combustion device complete combustion is achieved resulting in water, carbon dioxide and trace levels of ash. Recovery of polystyrene via composting or biodegradation is not possible. To find out more about polystyrene recycling, click here.
|







